Coming Together
Another question naturally follows the one about elements: “What makes two elements stick together?”
That simple query opens the next chapter of Coherent Wonder. This time, we don’t just see the atoms’ inner workings — we watch them converge, drawn by irresistible attraction into shared motion. Through new Coherence GeometryTM simulations, orbitals stretch, connect, and pull together with astonishing precision, each adjusting to the other until their regions of highest attraction intensify. What begins as curiosity turns into wonder — the moment when two entities remain themselves, yet simultaneously become an inseparable one.
A Deeper Look
When two atoms approach, we don’t see a collision. We see migration.
In these simple Coherence GeometryTM hydrogen molecule simulations, clouds of electron intensity seem to sense each other’s presence long before they meet. When two independent hydrogen atoms come close enough, small regions within each cloud — like tiny magnetic domains — begin to shift, inching toward the space between them. It’s as if the entire structure reorganizes itself in anticipation of connection.
The dynamic process is mesmerizing. The electron clouds from the two atoms pierce each other’s boundary, stretching across the gap to grab hold of what might be the partner’s invisible Coulomb shell, as shown in Figure 2. It takes time — a delicate search for balance — and then, once a solid “grip” is established, more of the electron magnet army begins to follow.

Figure 1: An H atom cloud reaching over to grip the other’s Coulomb shell.
The migrating regions then appear to march into alignment along that bridge, reinforcing it from both sides until a bright, localized electron intensity builds between them. This endpoint is most likely what we could call a bond — a bright, self-stabilized belt formed from two once-separate systems that have found common ground.
This bonding evolution sequence is captured at six different time steps in Figure 2. The favorable final alignment could just be a lucky coincidence. However, it also looks remarkably like an intelligent, intuitive way for nature to join forces.
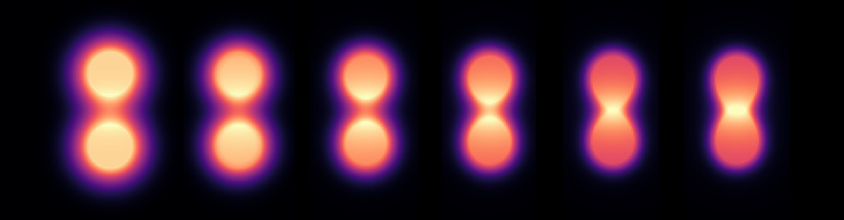
Figure 2: A simulatied H2 molecule forming in an intuitive, continuous process.
Hydrogen fluoride tells a different story. Here, the “smaller” Hydrogen electron cloud expands outward from its center, slipping into the larger atom’s nearest lobe and settling there — a resting place from which it apparently can’t escape.
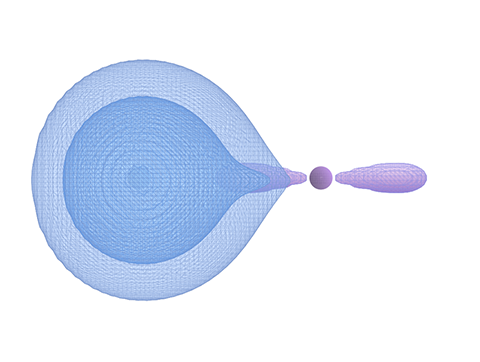
Figure 3: Hydrogen cloud reaches into fluorine lobe.
The connection isn’t forced; it’s simply a shape that fits — and holds. Meanwhile, back at base, the hydrogen Coulomb forces are holding tight, not letting the hydrogen cloud get pulled away. That tension forms a natural teardrop shape around the p-lobe, extending outward and physically changing the shape of the fluorine lobe in the process.
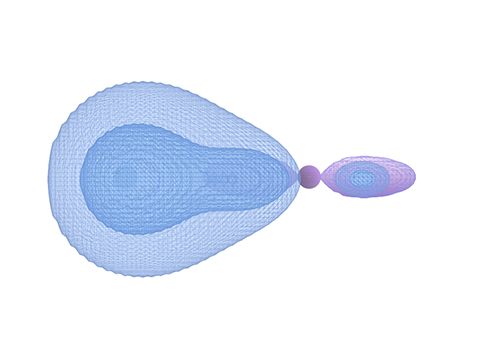
Figure 3: Final HF bonded state.
This kind of self-stabilization doesn’t look random to us. It looks deliberate — inevitable — a balanced alignment of competing forces. And again, we have to emphasize: we don’t tell these systems what to do. This isn’t AI hallucination; there’s no AI at all. These structures are self-forming and self-modulated. Just look at us — we’re as surprised as anyone, simply watching the scenes unfold.
We don’t pretend to know what’s really happening here. Maybe it’s nothing — or maybe Petersen’s Coherence GeometryTM is showing us something we haven’t quite understood yet. All we can say is that it happens, and it happens beautifully. We’ll keep exploring, and if it stirs your curiosity too, we’d love to have you wonder along with us.



